MTA-SZTE Biomimetic Systems Research Group
Principal investigator: Gábor Tóth
Projects
 The ion channels play extremely important role in the regulation of the living organisms. Their malfunction causes numerous serious diseases (circulatory problems, neurological disorders, kidney diseases, immunological problems.)
The ion channels play extremely important role in the regulation of the living organisms. Their malfunction causes numerous serious diseases (circulatory problems, neurological disorders, kidney diseases, immunological problems.)
Therefore, the understanding and controlling of their function are extremely important. A long standing research field of our research group is the synthesis and investigation of multiple disulfide containing peptides, including ion channel blockers. Recently, together with scientist from University of Debrecen, we got interesting results concerning the specific inhibition of the Kv 1.3 channel [Sci Rep 2015, 5, 18397., Chem Sci 2016, 7, 2666]. Continuing this cooperation, we plan to design and prepare further selective blockers for the Kv 1.3 and KCa 1.1 channels.
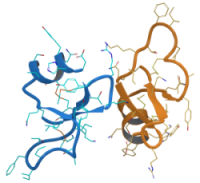
Antimicrobial and antibiotic resistances are among the biggest threats to global health. Estimates are that up to several million deaths result per year. Therefore the need for effective drug molecules enormously increasing. One of the most promising family are the antimicrobial peptides. There are two main groups of the known more than 3000 AMP:
- Linear, usually helical peptides (e.g. Cathelicidines)
- Cysteine rich, disulfide bridges containing peptides (e.g. defensins, NCR molecules, cysteine rich small antifungal proteines)
Recently, together with cooperating partners in Szeged and Debrecen we started to search peptides, which could serve as lead compounds for development antimicrobial agents in both families.
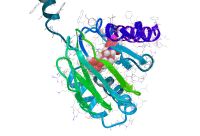
The sigma-1 receptor (Sig-1R), a well-known ER (endoplasmic reticulum)-chaperone localizes in the MAM (mitochondria-associated ER-membrane). It serves for Ca2+-signaling between the ER and mitochondria, and also acts as central modulator in inter-organelle signaling. It helps cell survival by attenuating ER-stress. Our goal is to find new selective Sig-1R agonist and investigate they mode of action. To this end we use the tools of computational drug design from virtual screening to molecular dynamics based methods. The selected compounds will be tested using in-vitro radioligand binding assay. Their neuroprotective effect via the suppression of ER stress will be tested on SH-SY5Y cell line.

Several biodegradable macromolecules such as polyesters, proteins, polysaccharides, polyelectrolytes, lipids or inorganic materials (layer double hydroxides (LDH), clays, mesoporous silica) are used as drug carriers in order to achieve a targeted drug delivery system and also a controlled drug release process. Nowadays, the application of core-shell nanoparticles (CSNPs) has become an area of intense growing interest. The albumin-based NPs are considered to be attractive opportunity as carrier systems because many binding sites are reachable to various drug molecules. The albumins have several specific advantages in nano-scale range, such as biodegradability, biocompatibility and non-toxicity. In our research group mainly albumin proteins as well as biocompatible polymers, hydrophobized hyaluronic acid and inorganic materials have been used for fabrication of CSNPs containing small drug molecules and the structure-dependent drug release processes are also studied.

Functionalized colloidal gold nanoparticles (Au NPs) and highly fluorescent gold nanoclusters (Au NCs) have been synthesized by biocompatible methods. Depending on the applied reducing and stabilizing agents (e.g. amino acids, peptides, large proteins or nucleotides) the fabricated nanosized hybrid systems (particles or clusters) exhibit plasmonic feature or intense fluorescence in the range of 450-700 nm, respectively. The synthesized gold nanohybrid systems show high kinetic stability at physiological conditions. The blue-, yellow-, orange-, or red-emitting gold nanoclusters have been used for development of selective and sensitive sensors for toxic or essential metal ions or small drug molecules. Moreover, the possibility of the fluorescence labelling of different drug-containing nanocarrier systems with the above mentioned sub-nanometer sized Au NCs has also been investigated.
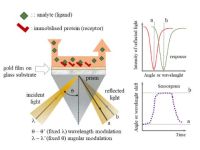
Quantitative, kinetic and thermodynamic characterization of receptor-ligand and protein-drug molecule interactions has been carried out by two-dimensional surface plasmon resonance (SPR) spectroscopy measurements. Investigation of temperature-dependent adsorption phenomena at solid/liquid interface permits the determination of thermodynamic state functions (ΔG, ΔH°, ΔS°) of the interaction. These results contribute to the better understanding the mechanism of these interactions. Several kinetic models are used for fitting the experimental sensograms. Moreover, the SPR spectroscopy is used to investigate protein-drug molecule and polymer-drug molecule interactions in order to optimize the synthesis of certain neurotransmitter and nonsteroidal anti-inflammatory drug molecules-containing protein- and biocompatible polymer-based nanocarrier composite systems.
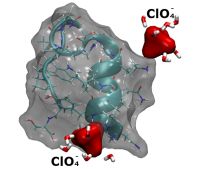
The fundamental influence of interfacial water on the macromolecules of biological systems can be modified by adding neutral salts to the solution (Hofmeister effect (HE)). Despite the widespread use of HE in colloid chemistry, preparative biochemistry and biotechnology, interpretation of the effects has remained a matter of debate. Recently, we published a theoretical grounding of the effects based on the salt dependence of solute-water interfacial tension. It was shown that the relation between the interfacial tension and protein structural stability is directly linked to protein conformational fluctuations. A methodological implication of the results is that HE is expected to change the reactions accompanying major conformational changes that involve water-exposed surface area changes of macromolecules and supramolecular assemblies. The aim of our project is to utilize HE as research tool to reveal the role of interfacial water structure in protein structure, protein-protein and protein-ligand interactions.

